Dr Eleanor Roberts
Mitochondria are small organelles found in large quantities in almost all cells of the body. They are thought to have once been independent entities that joined forces with our single-celled ancestors as each organism was advantageous to the other. This is suspected as mitochondria have their own DNA and can, independently from a cell’s nuclear DNA, produce some of the proteins needed for them to function. As mitochondria are found in egg cells in women but not the part of sperm cells that enters the egg during conception, we only inherit mitochondria from our mothers.
The main role of mitochondria is to produce energy stored in the bonds of the molecule adenosine triphosphate (ATP). In terms of ME/CFS, it’s interesting to understand some of the key components that lead to ATP generation so as to understand the implications of studies where these components are disrupted in people with ME/CFS, as well as the reason why some of the compounds utilised in energy metabolism pathway, such as nicotinamide, Coenzyme Q 10 (CoQ10) and acetyl-L-carnitine, are used by some with the goal of alleviating fatigue-related symptoms.
The structure of mitochondria
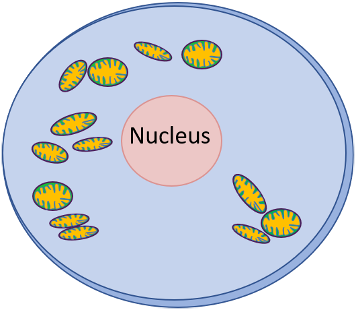
Mitochondria are around 0.5−1.0 micrometres (µm) in diameter, are mostly oblong shaped and have a two-layered cell membrane. The outside membrane is relatively smooth whereas the inner one is highly folded to extend its length, forming finger-like processes called cristae, where energy production is highest (Figure 1).
The more energy needed, such as in heart muscle, the more folded the inner membrane is. Additionally reflecting energy requirements, different parts of the body have different amounts of mitochondria, with high levels in not only the heart but also muscle, liver, kidney and brain cells.
What is energy?
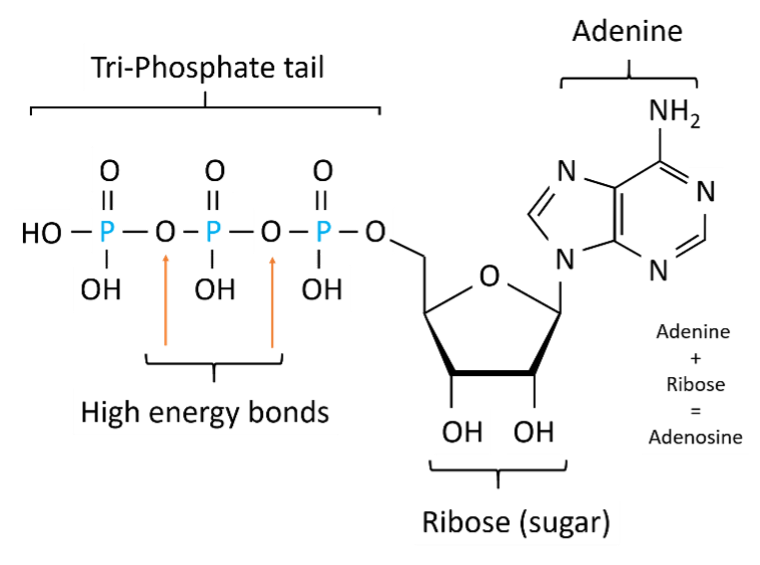
Energy is produced via cellular respiration in a process known as oxidative phosphorylation or OXPHOS, but before we go into how energy is made, it’s good to understand what exactly this energy is. Adenosine triphosphate (ATP) is a compound made up of the nucleotide adenine, a ribose sugar and three phosphates (Figure 2).
ATP creation arises when a phosphate group is added to the molecule adenosine diphosphate (ADP), which is almost the same molecule but with only two phosphate groups. This occurs through a process called oxidation where an electron is released via the gain of oxygen or loss of hydrogen. The opposite of oxidation is reduction, where electrons are gained via loss of oxygen or gain of hydrogen. This is a fundamental part of many of the chemical reactions that occur during ATP generation.
When the phosphate group is added to ADP, a high, negatively charged energy bond forms. This bond can be thought of like the type of cupboard door latch or a hair clip that is ‘clicked’ into place but springs back when released. In ATP, energy is ‘released’ by hydrolysis, where the chemical bond is broken by attraction to a water molecule.
How is energy produced in mitochondria?
Energy/ATP generation predominantly starts with consumption of carbohydrates, including sugar and starch, or release of fatty acid molecules, stored following fat consumption. At its most basic, in a process known as aerobic respiration, ATP generation occurs when the carbohydrate/sugar glucose (C6H12O6) is oxidised (oxygen [O2] added) to form ATP, water (H2O) and carbon dioxide (CO2): C6H12O6 + 6O2 = ~38 ATP + 6CO2 + 6H2O.
While from the above equation this process may look very simple, it actually involves a large array of molecules and pathways. In the first stage of energy generation, known as glycolysis, glucose is metabolised into a chemical called pyruvate. This occurs in the cell cytoplasm, outside of mitochondria. In times of high energy demand (above the ‘aerobic threshold’ or amount of oxygen the body can get into the mitochondria), glucose can be broken down anaerobically (without oxygen) with the end product being lactic acid instead of pyruvate, the presence of which can lead to muscle fatigue and pain. This pathway is much less efficient with regard to extracting energy from glucose than the aerobic one.
Once formed, pyruvate is broken down to create three molecules. The ‘acetyl’ group (CH3CO) (Figure 3B, bottom) is added to a molecule called coenzyme A (CoA) to become acetyl coenzyme A (acetyl-CoA). The hydrogen (Figure 3B, top right) is ‘carried’ into the next stage of energy production by another coenzyme called nicotinamide adenine dinucleotide (NAD+). The leftover CO2 is the waste product of this process that we eventually breath out.
Following its production, acetyl-CoA goes into a cyclical pathway known as the Krebs, citric acid or tricarboxylic acid cycle. This occurs in the matrix of mitochondria (Figure 1B). While again, it’s a very complicated pathway, at base, acetate (C2H3O−2) from acetyl-CoA is oxidised to form CO2 and H2O, with hydrogen ions (H+ also known as protons) being released to proton carriers such as NAD+ and flavin adenine dinucleotide (FAD). In this ‘reduced’ state, these carriers are known as NADH and FADH2.
NADH and FADH2 then enter the electron transport chain (ETC), the process at the heart of mitochondrial production of ATP. This takes place at the mitochondrial inner membrane (Figure 1B), predominantly in the cristae. The ETC is made up of a series of protein molecules, called Complexes I−IV, that carry electrons along in a defined sequence (Figure 4).

I−IV: Complexes; ADP: Adenosine diphosphate; ATP: Adenosine triphosphate; CoQ: Coenzyme Q; Cyt C: Cytochrome C; e-: electrons; FADH: Oxidised flavin adenine dinucleotide; FADH2: Reduced flavin adenine dinucleotide; H+: Protons; H: Hydrogen; H2O: Water; NADH: Reduced nicotinamide adenine dinucleotide; NAD+: Oxidised nicotinamide adenine dinucleotide; O2: Oxygen; Pi: Phosphate group
The ETC can be thought of like a pumping station of a hydroelectric dam where water is drawn into a pool up a hill using a small amount of energy then released at the top of the hill to cascade down and generate larger amounts of energy.
At the start of the ETC, two electrons (e−) are released from NADH into Complex I, making it ‘super charged.’ This gives it the energy to pump H+ from the matrix into the intermembrane space (Figure 1B), forming a gradient where there is more H+ outside the matrix then inside (Figure 4). These electrons are then passed to Coenzyme Q (CoQ). FADH2 also contributes electrons to CoQ, via Complex II. Together, these electrons are passed to Complex III, supercharging it and drawing more H+ across the membrane. The electrons then get passed along to Cytochrome C and into Complex IV, again supercharging it and pumping H+ from the matrix into the intermembrane space. In the final stage of the ETC, the electrons move from Complex IV back into the matrix, to be ‘accepted’ by O2, which is split, then joins with H+ to form water 2H2O, removing more free H+ from the matrix to create an even greater local H+ gradient.
As this pump system means there is now a glut of H+ in the intermembrane space, H+ want to flow back into the matrix to form an equilibrium between the two areas. They do this via a complex protein molecule in the inner membrane called ATP synthase (Figure 4). Here, like a water wheel driving an attached millstone, the H+ flow is used to drive a three chambered cylinder that picks up ADP and the phosphate group in the first third of its turn, brings them close enough together so they bond to form ATP in the second third, then releases ATP in the final third of its turn.
From one glucose molecule, there is usually enough of an H+ gradient generated to produce around 32−34 molecules of ATP. This can then be transported out of the mitochondria to places they are needed, such as for muscle contraction, cell division or cell function.
Resources
A more in depth look at mitochondria can be found in Cooper. The Cell, 2nd edition: https://www.ncbi.nlm.nih.gov/books/NBK9896/, with a more in depth look at cell respiration/oxidative phosphorylation here: https://www.ncbi.nlm.nih.gov/books/NBK9885/ and here: https://www.ncbi.nlm.nih.gov/books/NBK9903/.
More about ATP production can be found in this article: Bonora et al. ATP synthesis and storage. Purinergic Signalling 2012;8:343−57. https://bit.ly/3fSmpfs.
Read Eleanor’s second article on the mitochondria and ME/CFS